KC-1307
MC38-XH-Cell-Line
Home » MC38-XH-Cell-Line
Background of MC38-XH-Cell-Line
MC38 cell was derived from C57BL/6 mouse colorectal adenocarcinoma cell lines that can form tumors and
metastases after implantation in C57BL/6 mice or immunocompromised mice. The cell line was often used to
study the incidence and metastasis direction of colorectal cancer and became a common approach for efficacy
verification of tumor.
Specifications
| Catalog Number | KC-1307 |
|---|---|
| Cell Line Name | MC38-XH-Cell-Line |
| Host Cell Line | NA |
| Quantity | Two vials of frozen cells (≥2-106/vial) |
| Stability | Stable in culture over a minimum of 10 passages |
| Application | 3D cell culture; Cancer research |
| Freezing Medium | 70% DMEM+20% FBS+10% DMSO |
| Propagation Medium | DMEM+10%FBS |
| Selection Marker | NA |
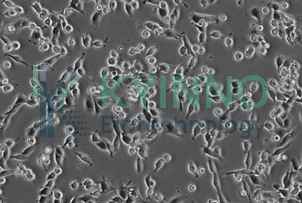
| Morphology | Adherent, fibroblast-like cells growing in monolayers |
| Subculture | Split saturated culture 1:10 every 3-4 days; seed out at about 1-3 × 105 cells/mL |
| Incubation | 37 °C with 5% CO2 |
| Storage | Liquid nitrogen immediately upon receiving |
| Doubling Time | Approximately 20 hours |
| Mycoplasma Status | Negative |
| In Vivo Validation | Yes |
Cell Line Generation
NA
Cell Resuscitation
- Prewarm culture medium (DMEM + 10% FBS)in a 37°C water bath.
- Thaw the frozen vial in a 37°C water bath for 1-2 minutes.
- Transfer the vial into biosafety cabinet, and wipe the surface with 70% ethanol.
- Unscrew the top of the vial and transfer the cell suspension gently into a sterile centrifuge tube containing 9.0mL complete culture medium.
- Spin at ~ 125 × g for 5-7 minutes at room temperature, and discard the supernatant without disturbing the pellet.
- Resuspend cell pellet with the appropriate volume of complete medium and transfer the cell suspension into a T25 culture flask.
- Incubate the flask at 37°C, 5% CO2 incubator.
- Split saturated culture 1:10 every 3-4 days; seed out at about 1-3 × 105 cells/mL.
Cell Freezing
- Prepare the freezing medium (70% DMEM + 20% FBS + 10% DMSO) fresh immediately before use.
- Keep the freezing medium on ice and label cryovials.
- Transfer cells to a sterile, conical centrifuge tube, and count the cells.
- Centrifuge the cells at 250×g for 5 minutes at room temperature and carefully aspirate off the medium.
- Resuspend the cells at a density of at least 3×106 cells/mL in chilled freezing medium.
- Aliquot 1 mL of the cell suspension into each cryovial.
- Freeze cells in the CoolCell freezing container overnight in a -80°C freezer.
- Transfer vials to liquid nitrogen for long-term storage
References
- PubMed=1149045 Corbett T.H., Griswold D.P. Jr., Roberts B.J., Peckham J.C., Schabel F.M. Jr.Tumor induction
- relationships in development of transplantable cancers of the colon in mice for chemotherapy assays, with a
- note on carcinogen structure.Cancer Res. 35:2434-2439(1975)
- PubMed=1255804; DOI=10.1093/jnci/56.4.871 Tan M.H., Holyoke E.D., Goldrosen M.H.Murine colon adenocarcinomas: methods for selective culture in vitro.J. Natl. Cancer Inst. 56:871-873(1976)