KC-1557
293T-mouse-CD137 Cell Line
Home » 293T-mouse-CD137 Cell Line
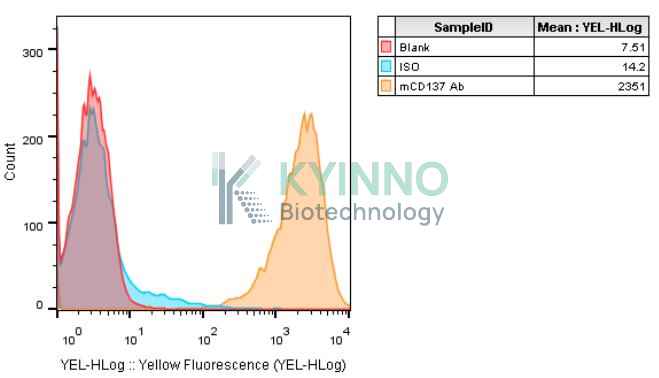
Background of 293T-mouse-CD137 Cell Line
CD137, also named 4-1BB ligand receptor and TNFRSF9, is a member of the tumor necrosis (TNF) receptor family and a famous co-stimulatory immune checkpoint molecule. CD137 is expressed on activated T cells, and nature killer cells, crosslinking of CD137 by agonistic antibody or its ligand enhance T cell proliferation, IL2 secretion and cytolytic activities.
Specifications
| Catalog Number | KC-1557 |
|---|---|
| Cell Line Name | 293T-mouse-CD137 Cell Line |
| Host Cell Line | 293T |
| Description | Stable 293T cell line expressing exogenous mouse-CD137 gene |
| Quantity | Two vials of frozen cells (≥2-106/vial) |
| Stability | Stable in culture over a minimum of 10 passages |
| Application | Drug screening and biological assays |
| Freezing Medium | 70% DMEM+20% FBS+10% DMSO |
| Propagation Medium | DMEM+10% FBS +1μg/mL Puromycin |
| Selection Marker | Puromycin |
| Morphology | Epithelial |
| Subculture | Split saturated culture 1:4-1:8 every 2-3 days; seed out at about 1-3 × 105 cells/mL |
| Incubation | 37 °C with 5% CO2 |
| Storage | Liquid nitrogen immediately upon receiving |
| Doubling Time | Approximately 30 hours |
| Mycoplasma Status | Negative |
| In Vivo Validation | NA |
Cell Line Generation
293T-mouse-CD137 cell line was generated using lentivirus expressing mouse-CD137 sequence.
Cell Resuscitation
- Prewarm culture medium (DMEM supplemented with 10% FBS, 1μg/mL Puromycin)in a 37°C water bath.
- Thaw the frozen vial in a 37°C water bath for 1-2 minutes.
- Transfer the vial into biosafety cabinet, and wipe the surface with 70% ethanol.
- Unscrew the top of the vial and transfer the cell suspension gently into a sterile centrifuge tube containing 9.0mL complete culture medium.
- Spin at ~ 125 × g for 5-7 minutes at room temperature, and discard the supernatant without disturbing the pellet.
- Resuspend cell pellet with the appropriate volume of complete medium and transfer the cell suspension into a T25 culture flask.
- Incubate the flask at 37°C, 5% CO2 incubator.
- Split saturated culture 1:4-1:8 every 2-3 days; seed out at about 1-3 × 105 cells/mL.
Cell Freezing
- Prepare the freezing medium (70% DMEM + 20% FBS + 10% DMSO) fresh immediately before use.
- Keep the freezing medium on ice and label cryovials.
- Transfer cells to a sterile, conical centrifuge tube, and count the cells.
- Centrifuge the cells at 250×g for 5 minutes at room temperature and carefully aspirate off the medium.
- Resuspend the cells at a density of at least 3×106 cells/mL in chilled freezing medium.
- Aliquot 1 mL of the cell suspension into each cryovial.
- Freeze cells in the CoolCell freezing container overnight in a -80°C freezer.
- Transfer vials to liquid nitrogen for long-term storage.
References
- Wolfl, M, J Kuball, W Y Ho, H Nguyen, T J Manley, M Bleakley, and P D Greenberg. 2007. “Activation-Induced Expression of CD137 Permits Detection, Isolation, and Expansion of the Full Repertoire of CD8+ T Cells Responding to Antigen Without Requiring Knowledge of Epitope Specificities.” Blood 110 (1). American Society of Hematology: 201–10. doi:10.1182/blood-2006-11-056168.
- Litjens, N H R, E A de Wit, C C Baan, and M G H Betjes. 2013. “Activation-Induced CD137 Is a Fast Assay for Identification and Multi-Parameter Flow Cytometric Analysis of Alloreactive T Cells.” Clinical and Experimental Immunology 174 (1): 179–91. doi:10.1111/cei.12152.