KC-1984
Jurkat-NFAT-Luc2-PVRIG
Home » Jurkat-NFAT-Luc2-PVRIG
Background of Jurkat-NFAT-Luc2-PVRIG
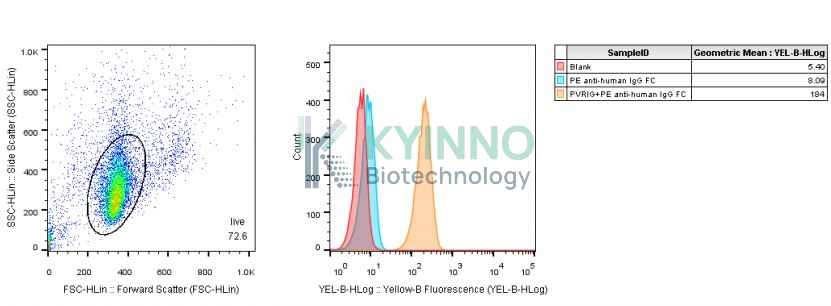
PVRIG, also known as CD112R, is a transmembrane protein that can act as a coinhibitory molecule to suppress Tcell receptor mediated signals. Studies have already shown that a PVRIG gene can inhibit CD8+ T cell function, thus making it a potential novel checkpoint for human T cells.
NFAT proteins, which are expressed in most immune-system cells, play a pivotal role in the transcription of cytokine genes and other genes critical for the immune response. Nuclear factor of activated T cells (NFAT), which is the pharmacological target of immunosuppressants cyclosporine and tacrolimus, has been shown to play an important role not only in T cells (immune system), from which their ame is derived, but also in many biological events. The activity of NFAT proteins is tightly regulated by the calcium/calmodulin-dependent phosphatase calcineurin, a primary target for inhibition by cyclosporin A and FK506. Calcineurin controls the translocation of NFAT proteins from the cytoplasm to the nucleus of activated cells by interacting with an N-terminal regulatory domain conserved in the NFAT family. The DNA-binding domains of NFAT proteins resemble those of Rel-family proteins, and Rel and NFAT proteins show some overlap in their ability to bind to certain regulatory elements in cytokine genes. NFAT is also notable for its ability to bind cooperatively with transcription factors of the AP-1 (Fos/Jun) family to composite NFAT: AP-1 sites, found in the regulatory regions of many genes that are inducibly transcribed by immune-system cells.
Specifications
| Catalog Number | KC-1984 |
|---|---|
| Cell Line Name | Jurkat-NFAT-Luc2-PVRIG |
| Host Cell Line | Human Jurkat cell line |
| Description | Jurkat cell line stable expressing exogenous luciferase gene under the control of NFAT responsive element and full-length human PVRIG protein |
| Quantity | Two vials of frozen cells (≥2-106/vial) |
| Stability | Stable in culture over a minimum of 10 passages |
| Application | Drug screening and biological assays |
| Freezing Medium | 70% RPMI1640+20% FBS+10% DMSO |
| Propagation Medium | RPMI1640+10%FBS+300µg/mL Hygromycin B+0.75µg/mL Puromycin |
| Selection Marker | Puromycin |
| Morphology | Lymphoblast |
| Subculture | Split saturated culture 1:4-1:6 every 2-3 days; seed out at about 1-3 × 105 cells/mL |
| Incubation | 37 °C with 5% CO2 |
| Storage | Liquid nitrogen immediately upon receiving |
| Doubling Time | Approximately 26 hours |
| Mycoplasma Status | Negative |
| In Vivo Validation | NA |
Cell Line Generation
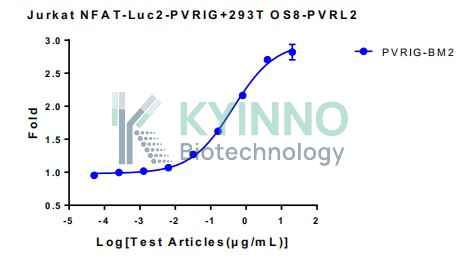
Jurkat NFAT-Luc2-PVRIG reporter cell line was generated using a lentiviral vector expressing PVRIG full-length sequence.
Cell Resuscitation
- Prewarm culture medium (RPMI1640 + 10% FBS + 300µg/mL Hygromycin B + 0.75µg/mL Puromycin) in a 37°C water bath.
- Thaw the frozen vial in a 37°C water bath for 1-2 minutes.
- Transfer the vial into biosafety cabinet, and wipe the surface with 70% ethanol.
- Unscrew the top of the vial and transfer the cell suspension gently into a sterile centrifuge tube containing 9.0mL complete culture medium.
- Spin at ~ 125 × g for 5-7 minutes at room temperature, and discard the supernatant without disturbing the pellet.
- Resuspend cell pellet with the appropriate volume of complete medium and transfer the cell suspension into a T25 culture flask.
- Incubate the flask at 37°C, 5% CO2 incubator.
- Split saturated culture 1:4-1:6 every 2-3 days; seed out at about 1-3 × 105 cells/mL.
Cell Freezing
- Prepare the freezing medium (70% RPMI-1640 + 20% FBS + 10% DMSO) fresh immediately before use.
- Keep the freezing medium on ice and label cryovials.
- Transfer cells to a sterile, conical centrifuge tube, and count the cells.
- Centrifuge the cells at 250×g for 5 minutes at room temperature and carefully aspirate off the medium.
- Resuspend the cells at a density of at least 3×106 cells/mL in chilled freezing medium.
- Aliquot 1 mL of the cell suspension into each cryovial.
- Freeze cells in the CoolCell freezing container overnight in a -80°C freezer.
- Transfer vials to liquid nitrogen for long-term storage
References
- Rao A, Luo C, Hogan PG. Transcription factors of the NFAT family: regulation and function. Annu Rev Immunol. 1997;15:707-47.
- Kitamura N, Kaminuma O. Isoform-Selective NFAT Inhibitor: Potential Usefulness and Development. Int J Mol Sci. 2021 Mar 8;22(5):2725.
- Sana I, Mantione ME, Angelillo P, Muzio M. Role of NFAT in Chronic Lymphocytic Leukemia and Other B-Cell Malignancies. Front Oncol. 2021 Apr 1;11:651057.