KC-3065
K562-HLA-A1101-Cell-Line
Home » K562-HLA-A1101-Cell-Line
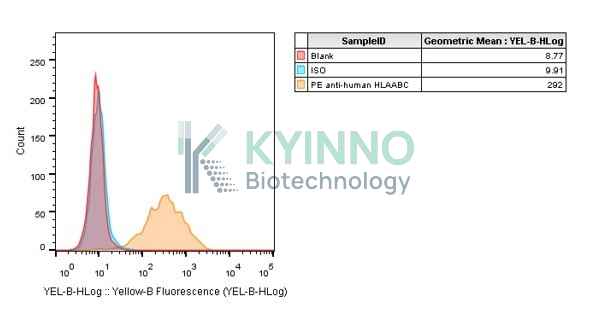
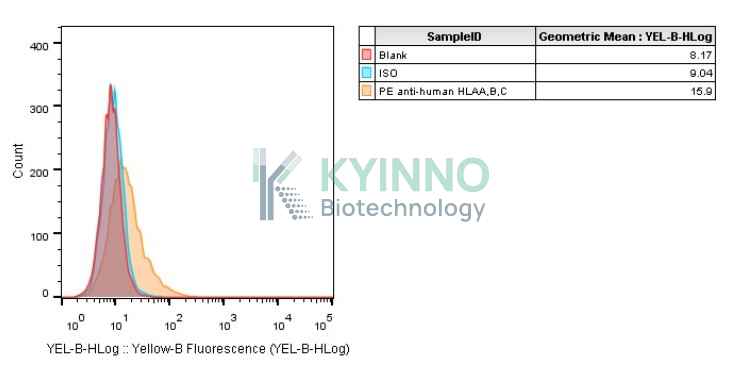
Background of K562-HLA-A1101-Cell-Line
HLA-A belongs to the HLA class I heavy chain paralogues. HLA-A is a heterodimer consisting of a heavy chain and a
light chai. The heavy chain is anchored in the membrane. HLA-A plays a central role in the immune system by
presenting peptides derived from the endoplasmic reticulum lumen so that they can be recognized by cytotoxic T
cells. They are expressed in nearly all cells. HLA-A is a Protein Coding gene. Diseases associated with HLA-A include
Severe Cutaneous Adverse Reaction and Birdshot Chorioretinopathy. Among its related pathways are SARS-CoV-2
Infection and Infectious disease.
Specifications
| Catalog Number | KC-3065 |
|---|---|
| Cell Line Name | K562-HLA-A1101-Cell-Line |
| Host Cell Line | K562 |
| Description | Stable K562 cell line expressing exogenous HLA-A1101 gene |
| Quantity | Two vials of frozen cells (≥2-106/vial) |
| Stability | Stable in culture over a minimum of 10 passages |
| Application | Drug screening and biological assays |
| Freezing Medium | 70% RPMI1640 + 20% FBS + 10% DMSO |
| Propagation Medium | RPMI1640 + 10% FBS + 300µg/mL Hygromycin B |
| Selection Marker | Hygromycin B |
| Morphology | Lymphoblast |
| Subculture | Split saturated culture 1:4-1:8 every 2-3 days; seed out at about 1-2 × 105 cells/mL |
| Incubation | 37 °C with 5% CO2 |
| Storage | Liquid nitrogen immediately upon receiving |
| Doubling Time | Approximately 20 hours |
| Mycoplasma Status | Negative |
| In Vivo Validation | NA |
Cell Line Generation
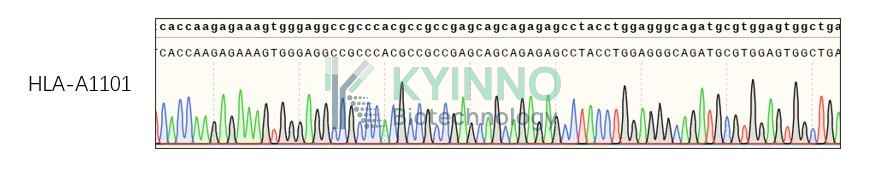
K562-HLA-A1101 Cell Line was generated using a lentiviral vector expressing the HLA-A1101 sequence.
Cell Resuscitation
1. Prewarm culture medium (RPMI164 supplemented with 10% FBS and 300µg/mL Hygromycin B)in a 37°C water bath.
2. Thaw the frozen vial in a 37°C water bath for 1-2 minutes.
3. Transfer the vial into biosafety cabinet, and wipe the surface with 70% ethanol.
4. Unscrew the top of the vial and transfer the cell suspension gently into a sterile centrifuge tube containing 9.0mL complete culture medium.
5. Spin at ~ 125 × g for 5-7 minutes at room temperature, and discard the supernatant without disturbing the pellet.
6. Resuspend cell pellet with the appropriate volume of complete medium and transfer the cell suspension into a T25 culture flask.
7. Incubate the flask at 37°C, 5% CO2 incubator.
8. Split saturated culture 1:4-1:8 every 2-3 days; seed out at about 1-2 × 105 cells/mL.
2. Thaw the frozen vial in a 37°C water bath for 1-2 minutes.
3. Transfer the vial into biosafety cabinet, and wipe the surface with 70% ethanol.
4. Unscrew the top of the vial and transfer the cell suspension gently into a sterile centrifuge tube containing 9.0mL complete culture medium.
5. Spin at ~ 125 × g for 5-7 minutes at room temperature, and discard the supernatant without disturbing the pellet.
6. Resuspend cell pellet with the appropriate volume of complete medium and transfer the cell suspension into a T25 culture flask.
7. Incubate the flask at 37°C, 5% CO2 incubator.
8. Split saturated culture 1:4-1:8 every 2-3 days; seed out at about 1-2 × 105 cells/mL.
Cell Freezing
1. Prepare the freezing medium (70% RPMI164 + 20% FBS + 10% DMSO) fresh immediately before use.
2. Keep the freezing medium on ice and label cryovials.
3. Transfer cells to a sterile, conical centrifuge tube, and count the cells.
4. Centrifuge the cells at 250×g for 5 minutes at room temperature and carefully aspirate off the medium.
5. Resuspend the cells at a density of at least 3×106 cells/mL in chilled freezing medium.
6. Aliquot 1 mL of the cell suspension into each cryovial.
7. Freeze cells in the CoolCell freezing container overnight in a -80°C freezer.
8. Transfer vials to liquid nitrogen for long-term storage
2. Keep the freezing medium on ice and label cryovials.
3. Transfer cells to a sterile, conical centrifuge tube, and count the cells.
4. Centrifuge the cells at 250×g for 5 minutes at room temperature and carefully aspirate off the medium.
5. Resuspend the cells at a density of at least 3×106 cells/mL in chilled freezing medium.
6. Aliquot 1 mL of the cell suspension into each cryovial.
7. Freeze cells in the CoolCell freezing container overnight in a -80°C freezer.
8. Transfer vials to liquid nitrogen for long-term storage
References
1. Arce-Gomez B, Jones EA, Barnstable CJ, Solomon E, Bodmer WF (February 1978). The genetic control of HLA-A
and B antigens in somatic cell hybrids: requirement for beta2 microglobulin. Tissue Antigens. 11 (2): 96–112.
doi:10.1111/j.1399-0039.1978.tb01233.x. PMID 77067.
2. de Campos-Lima PO, Levitsky V, Brooks J, et al. (April 1994). T cell responses and virus evolution: loss of HLA A11-restricted CTL epitopes in Epstein-Barr virus isolates from highly A11-positive populations by selective mutation of anchor residues. J. Exp. Med. 179 (4): 1297–305. doi:10.1084/jem.179.4.1297. PMC 2191457. PMID 7511684.
2. de Campos-Lima PO, Levitsky V, Brooks J, et al. (April 1994). T cell responses and virus evolution: loss of HLA A11-restricted CTL epitopes in Epstein-Barr virus isolates from highly A11-positive populations by selective mutation of anchor residues. J. Exp. Med. 179 (4): 1297–305. doi:10.1084/jem.179.4.1297. PMC 2191457. PMID 7511684.