康源博创抗体生产平台:高质量抗体开发的可靠合作伙伴
抗体发现服务
抗体产品概览
在 康源博创抗体生产平台,我们专注于为早期药物发现和临床前研究提供高质量抗体的开发与生产服务。依托优化的哺乳动物细胞表达系统,我们可基于 HEK293/CHO 细胞,为客户提供全面的抗体表达与生产服务。
从抗体序列信息出发,我们为客户提供涵盖基因构建、抗体表达、纯化、质量检测及交付的一站式解决方案,助力获得高纯度、低内毒素水平的优质抗体产品。

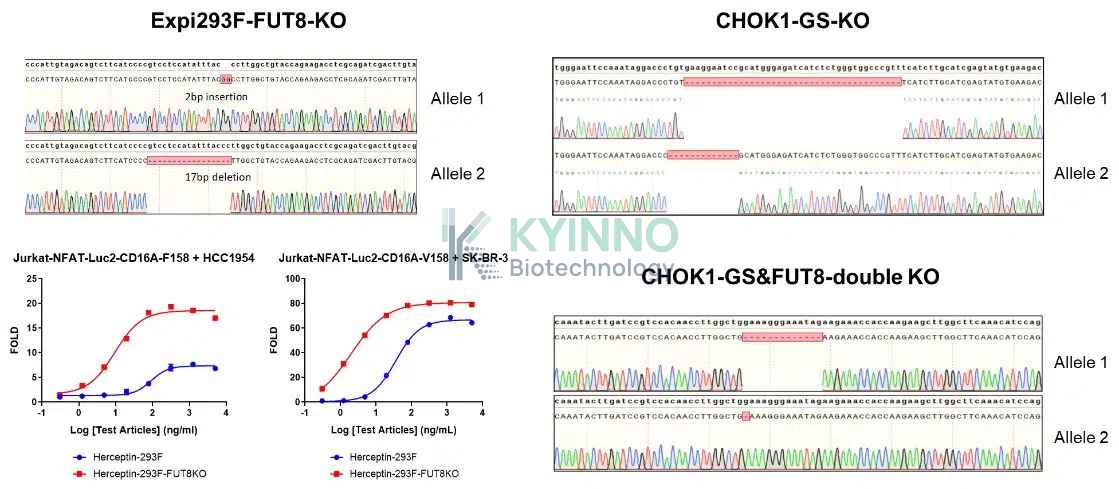
我们的抗体表达采用哺乳动物细胞表达系统,支持瞬时小规模表达,也可通过稳定转染构建大规模表达细胞株。瞬时转染系统包括 ExpiCHO-S、Expi-293F 及相应的 FUT8 敲除系统,可有效提升抗体依赖性细胞介导的细胞毒作用(ADCC)活性。此外,稳定转染细胞株表达系统采用 CHO-K1 细胞,并结合 GS 敲除系统及 GS-FUT8 双敲除系统。这些表达系统均经过精心筛选,以确保产品质量和产量达到最优水平。
KYINNO 抗体生产平台配备完善的硬件设施,包括生物反应器、5 套 AKTA 蛋白纯化系统,以及多种质量检测设备,如 Fortebio、流式细胞仪、HPLC、CE-SDS 和 LC-MS 等。
我们提供从抗体生产到质量检测的全流程服务。同时,产品储存缓冲液也可根据客户特定的制剂需求进行定制。
KYINNO 抗体生产平台可基于抗体序列信息,从基因构建与表达开始,为客户提供完整的抗体生产服务。依托高效、快速的重组抗体表达体系,我们可通过 CHO/HEK293 细胞转染,在最短 2 周内交付纯化抗体,并根据客户下游应用需求,满足高产量和高浓度的抗体制备要求。
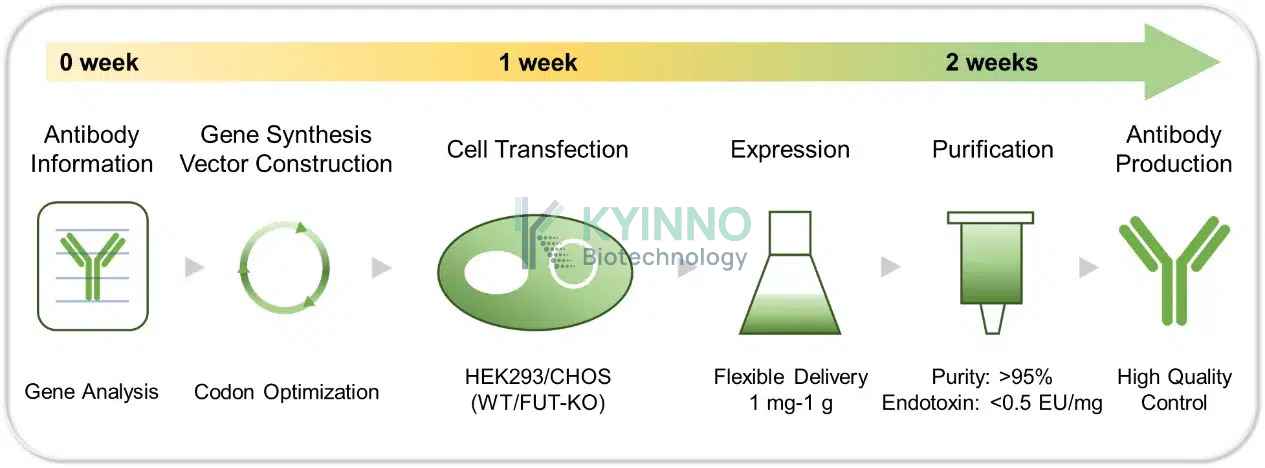
我们提供从抗体生产到质量检测的全流程服务。同时,产品储存缓冲液可根据客户特定的制剂需求进行定制。
康源博创抗体生产平台可基于客户提供的抗体序列信息,从基因构建与表达开始,提供一站式抗体开发与生产服务。依托高效、快速的重组抗体表达体系,我们可通过 CHO/HEK293 细胞转染,在最短 2 周内交付纯化抗体,并可根据客户下游应用需求,满足高产量和高浓度的抗体制备要求。
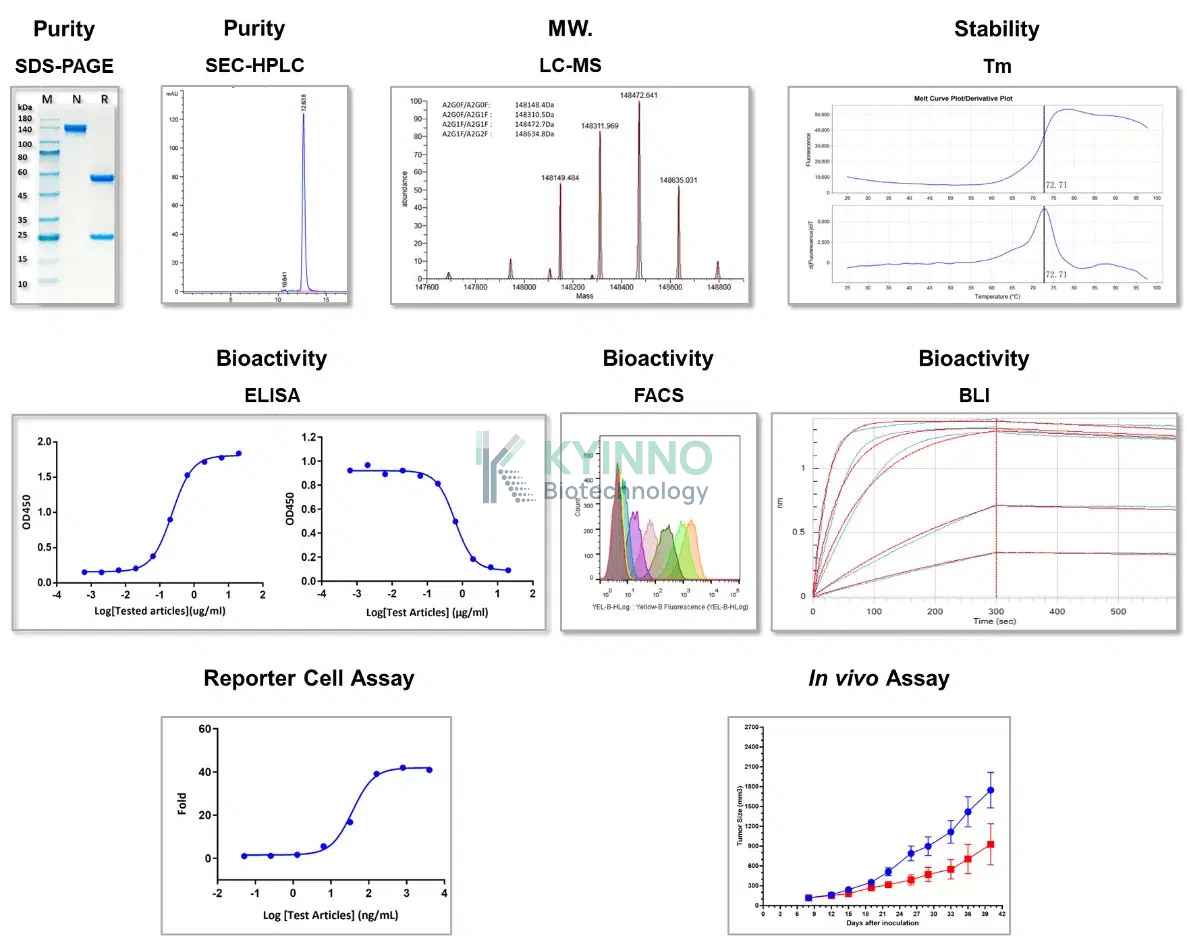
康源博创建立了严格的质量控制体系,并在大规模抗体生产方面积累了丰富的成功经验。我们的产品均经过严格检测,包括分子量分析、纯度评估、稳定性分析、亲和力和生物活性验证,以及功能实验和体内实验验证。
我们具备培养工艺和生产工艺优化能力,可满足大规模抗体生产需求,抗体生产规模覆盖毫克级至克级,并符合 内毒素 <0.5 EU/mg 的标准要求。
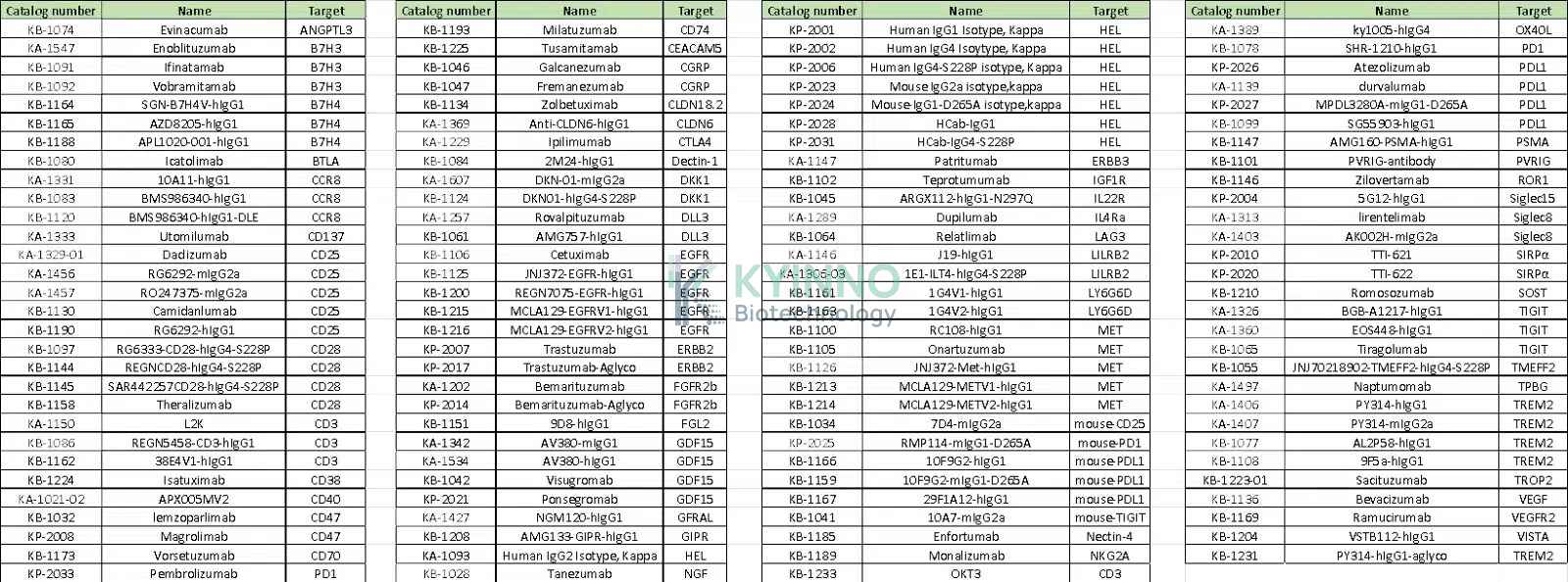
目前,KYINNO 现货库存涵盖 100 种 Benchmark 抗体,包括 20 余种带有 Fc 修饰的人源 IgG 同种型抗体,以及针对多种抗原靶点的对照抗体。每一款抗体均经过全面、严格的质量控制检测,确保产品质量稳定可靠。
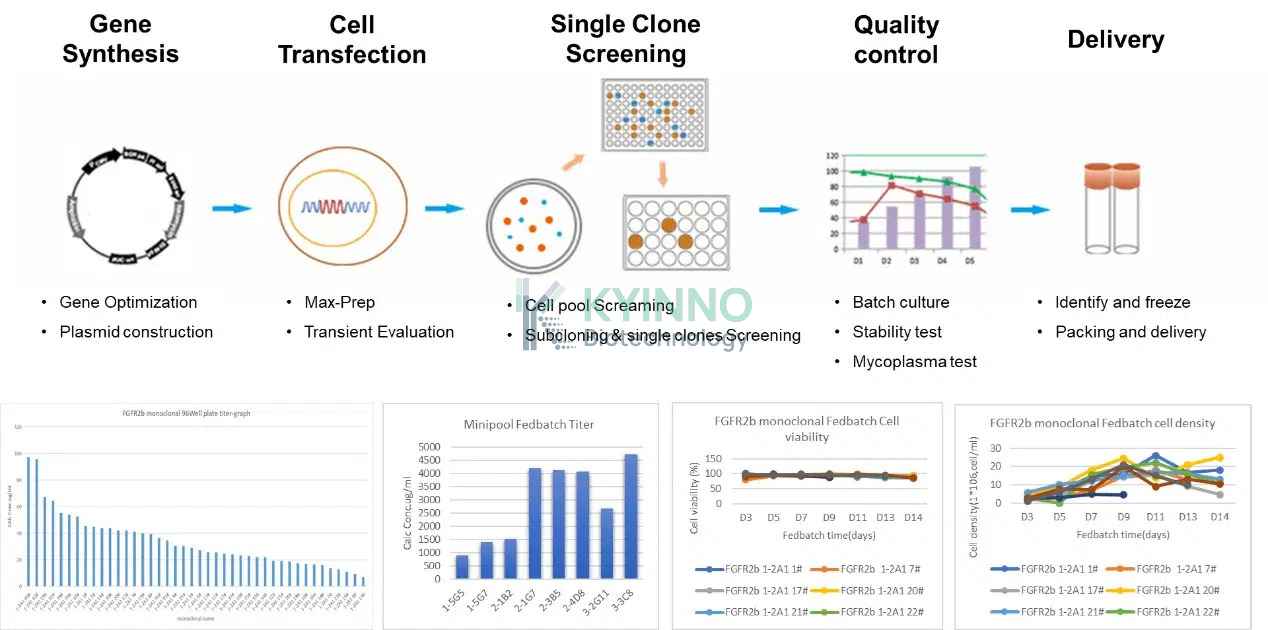
除标准抗体生产服务外,我们还可提供抗体表达稳定转染细胞株构建服务。该服务涵盖基因合成、细胞转染、单克隆筛选、严格质量评估,并最终交付符合要求的抗体表达稳定转染细胞株。
截至目前,KYINNO 抗体生产平台已成功构建 20 余种抗体稳定转染细胞株。
所有抗体发现服务
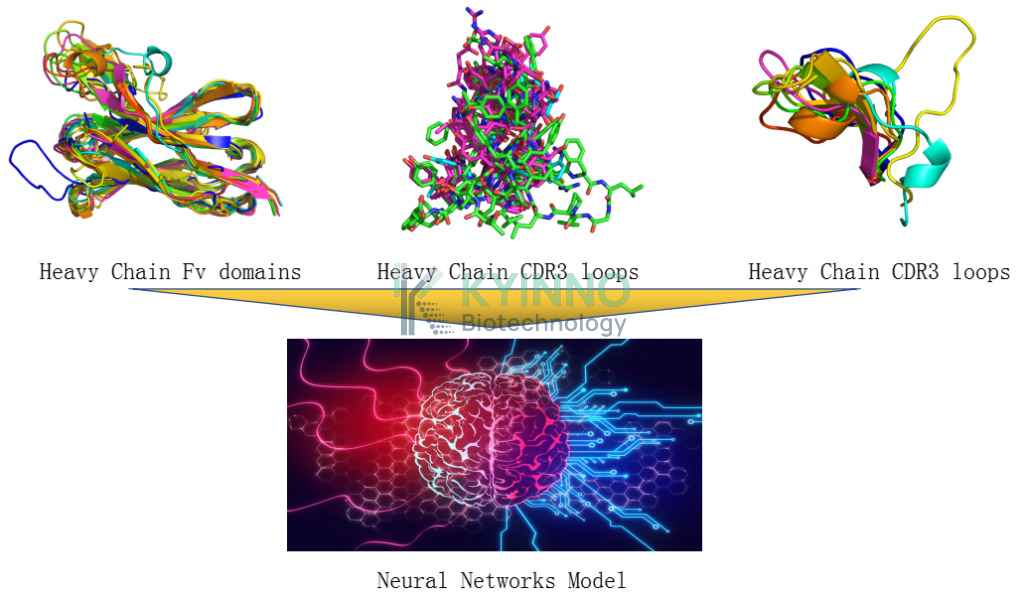
AI 药物发现服务
AI 药物发现利用先进算法,优化并重塑治疗性抗体开发流程,有效提升传统复杂研发过程的效率。我们的 AI 驱动神经网络能够精准识别抗体与抗原结构之间的关系,从而实现创新型抗体药物的虚拟生成与设计。
FAQs About Our Customized Antibody Production Service
What is the difference between antibody discovery and antibody production at KYINNO?
- Antibody Discovery (or “Custom Antibody Service”) involves generating novel antibody sequences against specific targets.
- Antibody Production (KYINNO’s focus) involves taking an existing or discovered antibody sequence and scaling it up—expressing, purifying, and delivering high-quality recombinant antibodies in the required quantities.
What expression systems do you use for antibody manufacturing?
- We primarily use mammalian cell expression systems, including HEK293 (e.g., Expi293) and CHO (e.g., ExpiCHO and CHOK1) cells.
- Depending on your needs, we can employ FUT8 knockout systems for enhanced ADCC and GS knockout systems (including GS-FUT8 double knockout) for optimized yields and product quality.
Can you produce antibodies at both small and large scales?
- Yes. Our platform supports milligram-scale production for early R&D phases and gram-scale production for preclinical applications. This flexibility ensures seamless scalability as your project moves forward.
How long does the antibody production process typically take?
- Production timelines vary based on project scope. For small-scale transient expression, we can deliver purified antibodies in as few as 2 weeks. For more extensive projects or stable cell line generation, timelines may be longer.
What quality control measures are in place?
- We have a stringent, multi-step QC workflow that includes:
- -Purity assessment via SDS-PAGE and SEC-HPLC
- – Molecular weight confirmation via LC-MS/MS.
- – Stability analysis.
- – Affinity and bioactivity validation (e.g., Fortebio, Biacore and flow cytometry).
- – Endotoxin testing (<0.5 EU/mg).
- – In vivo functional assessments when necessary.
Can I request a specific buffer or formulation for my antibodies?
- Absolutely. We offer customizable buffer formulations to accommodate your unique downstream application needs, whether it’s for further development, assay work, or preclinical studies.
Do you offer stable transfection cell line development?
- Yes. Beyond transient expression, KYINNO can build stable transfection cell lines, which involves gene synthesis, cell transfection, monoclonal screening, and thorough QC to ensure reliable, long-term antibody expression.
What level of endotoxin control do you maintain?
- We adhere to a strict endotoxin standard of <0.5 EU/mg, making our antibodies suitable for in vivo experiments and advanced drug development applications.
Do you offer additional services related to antibody production?
- In addition to manufacturing services, we also provide:
- Custom isotype switching and Fc modifications.
- Large-scale production (milligrams to grams).
- Assistance with technology transfer for your in-house production.
- Access to our inventory of 100 “Benchmark” antibodies with diverse Fc modifications.
How do I get started or request a quote?
- Simply reach out via our contact details or online inquiry form. Provide us with your antibody sequence information and project requirements. Our technical experts will work with you to develop a customized production plan.