| Catalog Number | KC-1637 |
|---|---|
| Cell Line Name | Ba/F3-TPR-Met-F1200I-Cell-Line |
| Host Cell Line | Ba/F3 |
| Description | Stable Ba/F3 clone expressing exogenous Met-F1200I mutation with TPR fusion . |
| Quantity | Two vials of frozen cells (≥2-106/vial) |
| Stability | Stable in culture over a minimum of 10 passages |
| Application | Drug screening and biological assays |
| Freezing Medium | 70% RPMI1640+20% FBS+10% DMSO |
| Propagation Medium | RPMI1640+10%FBS |
| Selection Marker | Puromycin |
| Morphology | Mostly single, round (some polymorph) cells in suspension |
| Subculture | Split saturated culture 1:10 every 3 days; seed out at about 1-3 × 105 cells/mL |
| Incubation | 37 °C with 5% CO2 |
| Storage | Liquid nitrogen immediately upon receiving |
| Doubling Time | Approximately 20 hours |
| Mycoplasma Status | Negative |
| In Vivo Validation | NA |

Figure: Characterization of Met and its mutants overexpressing in Ba/F3 stable clones using western blot.
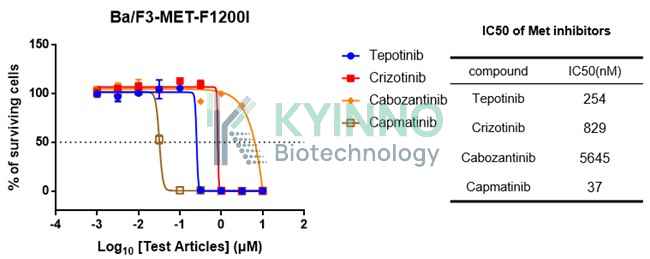
a. Cell-based kinase inhibition screening b. Cell viability assay c. In vivo efficacy study Example: kinase inhibitors screening 1. Harvest and seed the Ba/F3 cells expressing the MET mutant in a 96-well plate (3000 cells/90ul medium). 2. Add 10ul 10X serially diluted compound solution to each well the following day and incubate the plate for another 72 hours. 3. Add 100ul Cell Titer-Glo to each well, mix well and perform the analysis using Envision. 4. Plot the dose-responsive curve and fit the IC50 value (the centration of 50% inhibition of DMSO vehicle treated clones) using the GraphPad Prism software (Version 7).