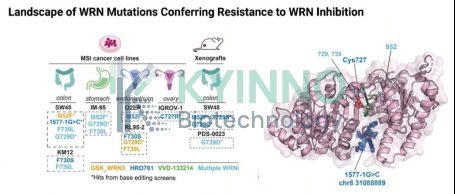
Introduction
At the AACR special session “Expanding and Translating Cancer Synthetic Vulnerabilities,” the team led by Faith C. Fowler presented recent findings showing that MSI-H cells, upon long-term exposure to HRO761 and two novel WRN inhibitors, develop multiple point mutations localized within the WRN helicase domain. These mutations directly disrupt inhibitor binding, thereby driving the emergence of drug resistance. This highlights an urgent need to develop second-generation WRN inhibitors capable of overcoming resistance, along with corresponding experimental models. Based on these insights, we established the WRN-MutCell platform, which employs CRISPR/Cas9 technology to precisely introduce distinct resistance-associated mutations into the endogenous WRN gene. This platform is designed to provide powerful tools for the molecular optimization and functional validation of next-generation WRN inhibitors.
About the WRN Target
Werner syndrome protein (WRN) is a key DNA helicase of the RecQ family, playing critical roles in replication fork restart, maintenance of genome stability, and protection of telomere function. In MSI-H tumors, defects in mismatch repair lead to the accumulation of aberrant DNA secondary structures, creating a pronounced dependence on WRN helicase activity. Inhibition of WRN function can therefore trigger a synthetic lethal effect, selectively eliminating MSI-H tumor cells.
Biological Functions
· Replication Fork Maintenance
In BRCA2-deficient cells, WRN relies on its ATPase and helicase activities to promote replication fork restart and to suppress MRE11-mediated fork degradation, thereby preserving genome stability.
· Helicase Core Structure
The helicase core region of WRN comprises the winged-helix (WH) domain and the ATP-binding pocket. Residue I852 is located within a critical α-helix; the I852F mutation alters enzymatic activity and can recapitulate resistance-associated phenotypes.
Disease Relevance
· Synthetic Lethal Dependency in MSI-H Tumors
WRN exhibits a synthetic lethal dependency in multiple MSI-H solid tumors, including colorectal cancer, gastric adenocarcinoma, and endometrial cancer, establishing it as an emerging target for precision oncology.
· Drug Sensitivity and Combination Strategies
Inhibition or loss of WRN markedly enhances the sensitivity of MSI-H tumors to DNA damage response–targeting agents, such as PARP inhibitors, providing a strong mechanistic rationale for the development of combination therapeutic strategies.
Current Development Landscape
· HRO761 (Novartis)
The first non-covalent WRN inhibitor to enter clinical development. It is currently in a Phase I clinical trial (NCT05838768), primarily targeting MSI-H colorectal cancer and other MSI-H solid tumors.
· NTX-452 (Nimbus Therapeutics)
A next-generation non-covalent WRN inhibitor that has demonstrated strong synthetic-lethal activity in preclinical models. Clinical initiation is planned for the first half of 2025.
· VVD-133214
A covalent, allosteric WRN inhibitor identified through a chemoproteomics-based strategy. It has advanced to the clinical candidate stage.
· RO7589831
A WRN inhibitor program supported by the U.S. National Cancer Institute (NCI), with a clinical development focus on multiple MSI-H solid tumors.
Summary
The HCT116-WRN-I852F-KI cell line accurately recapitulates the WRN-dependent synthetic lethal phenotype observed in MSI-H tumors, making it a critical model for the study of WRN inhibitors. This cell line can be broadly applied to:
· Mechanistic Studies of Drug Resistance
Elucidating how WRN-associated mutations mediate loss of inhibitor efficacy, thereby facilitating the development of second-generation, resistance-overcoming WRN inhibitors.
· Novel Drug Screening and Evaluation
Providing a robust and reliable platform for pharmacodynamic validation and prioritization of WRN-targeting compounds.
· Optimization of Combination Strategies
Systematically assessing the combinatorial potential of WRN inhibitors with DNA damage response agents, such as PARP inhibitors, to accelerate the development of precision therapeutic strategies.
WRN-MutCell Platform: Products and Services
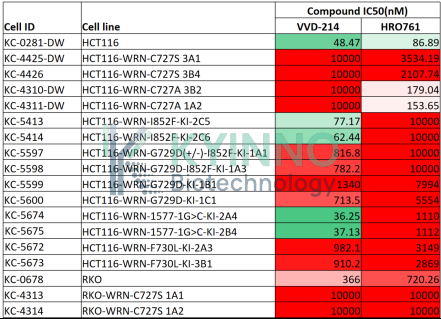
· HCT116-WRN-mut-KI Cell Line — In Vitro Validation Data
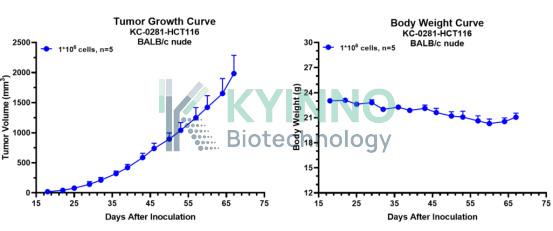
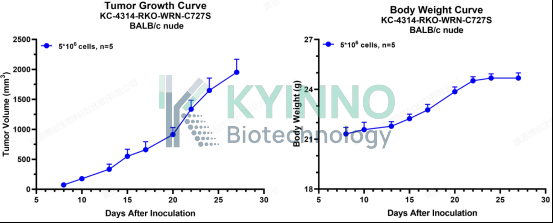
· HCT116-WRN-mut-KI Cell Line — In Vivo Validation Data
1、HCT116-WT
2、HCT116-WRN-C727A
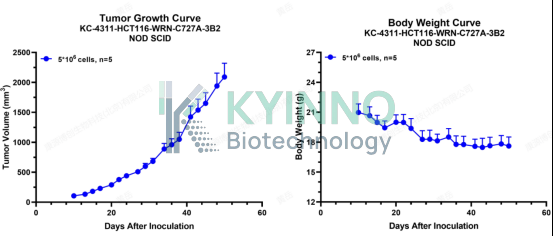 3、HCT116-WRN-C727A
3、HCT116-WRN-C727A
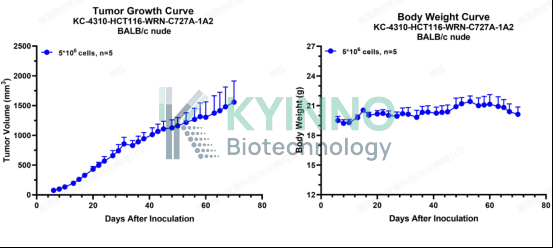
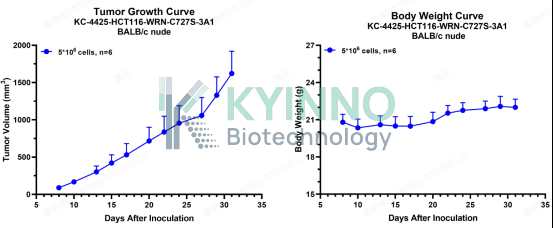
4、HCT116-WRN-C727S
5、RKO-WT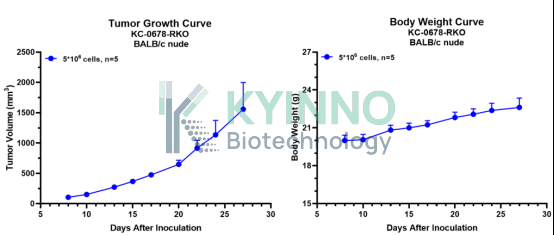
6、RKO-WRN-C727S
Abstract
We have recently developed a panel of HCT116-WRN-mutation cell lines by using CRISPR technology to precisely introduce multiple resistance-associated mutations into the endogenous WRN gene. Together with our previously established HCT116-WRN-C727S/A-KI and RKO-WRN-C727S-KI models, these cell lines constitute a comprehensive platform that faithfully recapitulates the synthetic lethal dependency of MSI-H tumors on WRN helicase activity.
This platform not only supports high-throughput drug screening, but also enables mechanistic dissection of resistance, as well as systematic evaluation of next-generation WRN inhibitors and combination therapeutic strategies.
