The “Golden Duo” of Cancer Immunotherapy: PD-1 & IL-2 Dual-Target Reporter Cells
PD-1: Core Mechanisms of Immune “Braking” and Therapeutic Limitations
The “golden duo” of tumor immunotherapy, PD-1 and IL-2, has once again captured global attention following the presentation of clinical data for Innovent’s PD-1/IL-2 dual-target therapy IBI363 at this year’s ASCO meeting, which directly led to a multi-billion-dollar strategic collaboration with Takeda and fully ignited enthusiasm for the PD-1/IL-2 dual-target field; as a flagship target in cancer immunotherapy, PD-1 has reshaped treatment outcomes for countless patients, yet its long-term benefit rate of only 20–30% has remained a challenging bottleneck, and the introduction of the classical immune cytokine IL-2 is now breaking this limitation through a synergistic “1+1 > 2” effect, for which Kyinno Biotechnology has purposefully developed PD-1/IL-2 dual-target reporter cell lines to enable efficient and precise evaluation of this synergistic immunotherapeutic potential.
IL-2: The Potential and Limitations of the Immune “Engine”
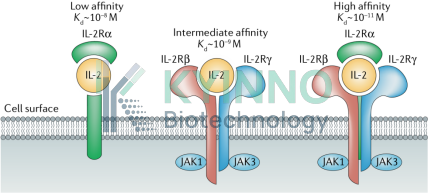
If PD-1 functions as an immune “brake,” then IL-2 serves as the “engine” that drives immune cell activation. As a key cytokine, IL-2 is mainly secreted by activated CD8⁺ T cells and exerts its functions through two types of receptors (Figure 1): the high-affinity trimeric receptor IL-2Rαβγ (CD25–CD122–CD132), which is expressed on tumor-specific T cells and regulatory T cells (Tregs) and can be activated by low doses of IL-2, and the intermediate-affinity dimeric receptor IL-2Rβγ (CD122–CD132), which is expressed on conventional effector T cells and requires high-dose IL-2 for activation. As early as 1990, recombinant IL-2 was approved for the treatment of melanoma and renal cell carcinoma, where it could induce durable tumor regression; however, its clinical application has been limited by an extremely short half-life of only a few minutes, the risk of severe toxicities such as vascular leak syndrome at high doses, and the potential activation of Tregs at low doses that can compromise therapeutic efficacy.
![]()
Figure 1. Binding of IL-2 to IL-2 Receptors with Different Affinities
Breaking the Bottleneck Together: The Core Rationale of PD-1 & IL-2 Dual Targeting
The core rationale for combining PD-1 and IL-2 lies in a precisely coordinated strategy of “release of inhibition plus strong activation,” with the STAT5 signaling pathway serving as the central hub linking these processes (Figure 2). This synergistic mechanism specifically targets progenitor exhausted T cells (Tpex) within the tumor microenvironment, which are rendered functionally dormant by PD-1 signaling. Inhibitory PD-1 signaling blocks STAT5 activation through the SHP2 phosphatase, a constraint that is lifted by PD-1 blockade; under these conditions, IL-2 selectively binds to the upregulated high-affinity trimeric receptor IL-2Rαβγ on Tpex cells, activating downstream JAK1/JAK3 kinases, driving STAT5 phosphorylation, dimerization, and nuclear translocation, and subsequently regulating the transcription of key target genes, thereby promoting the differentiation of Tpex into effector T cells with both potent cytotoxic activity and long-term persistence, while simultaneously counteracting Treg-mediated immunosuppression to establish a positive feedback loop of “pathway activation and functional reinforcement” that markedly reverses therapeutic resistance.
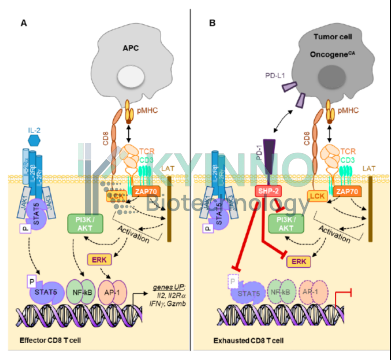
Figure 2. Mechanisms of Action of IL-2 and PD-1
Emerging Frontiers: Bispecific Molecules as the Mainstream Strategy
Current development has advanced from simple combination therapy to precise molecular integration, with bispecific molecules emerging as the dominant strategy. For example, Innovent’s IBI363 uses a PD-1–targeting arm to deliver IL-2 directly to the tumor site while structurally engineering IL-2 to enhance its selective binding to the high-affinity trimeric receptor; its phase I data are markedly superior to those of conventional IL-2 therapy and show efficacy even in PD-L1–negative patients. Roche’s RG6279 fuses a PD-1 antibody with an IL-2 variant to improve targeting through cis-activation, while Regeneron’s REGN10597 incorporates a masking design that keeps IL-2 inactive in circulation and only activates it upon PD-1 engagement in the tumor microenvironment. All of these innovative designs revolve around the same core goals of enhanced targeting and reduced toxicity, holding promise for breaking current therapeutic bottlenecks and bringing new hope to a broader population of cancer patients.
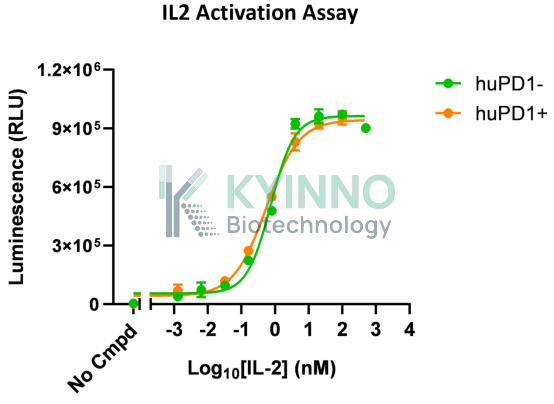
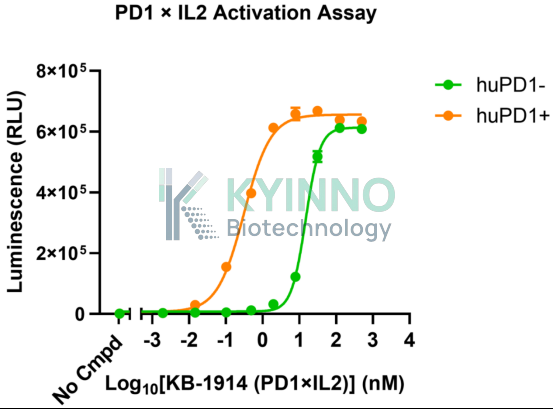
To address the R&D needs arising from the synergistic mechanism of PD-1 and IL-2 dual targeting, Kyinno Biotechnology has developed a reporter cell line, 293T-STAT5-Luc2-IL2RA-IL2RB-IL2RG-PD1 (validation data shown below). Its key advantage lies in the stable co-expression of PD-1 and the high-affinity trimeric IL-2 receptor IL-2Rαβγ, together with a STAT5-responsive Luc2 reporter gene. When PD-1 blockade relieves inhibitory signaling and IL-2 engages its receptor, activation of the STAT5 pathway drives Luc2 expression, allowing the efficiency of synergistic activation to be directly quantified through luminescence intensity.
Reporter Cell Product List:
|
KC-5588 |
293T-STAT5-Luc2-IL2RA-IL2RB-IL2RG |
|
KC-5667 |
293T-STAT5-Luc2-IL2RA-IL2RB-IL2RG-PD1 |
|
clones |
IL-2 EC50(nM) |
KB-1914[PD1×IL2] EC50(nM) |
|
huPD1- |
0.7438 |
13.79 |
|
huPD1+ |
0.3400 |
0.2622 |
This reporter cell line can be broadly applied to activity screening, potency assessment, and mechanism-of-action validation for PD-1/IL-2 bispecific antibodies, combination therapies, and other candidate agents, providing efficient and precise assay support to accelerate the translational development of PD-1 and IL-2 dual-target therapeutics. In addition to cell line development services, Kyinno Biotechnology also offers antibody discovery, antibody expression, off-target screening, and in vitro and in vivo pharmacology services, and we welcome you to contact us for more information.
1. References:
2. doi: 10.1038/s41577-018-0046-y
3. doi: 10.3390/cancers11121832
4. doi:10.1016/j.trecan.2023.11.008
5. 10.1158/2159-8290.CD-23-1263
6. 10.1016/j.xcrm.2024.101747
Abstract: PD-1 and IL-2 represent a “golden duo” in cancer immunotherapy, achieving a synergistic effect of “release of inhibition plus strong activation” through the STAT5 pathway and overcoming the limitations of monotherapy. Bispecific molecules have emerged as a major focus of current development. Kyinno Biotechnology has introduced the 293T-STAT5-Luc2-IL2RA-IL2RB-IL2RG-PD1 reporter cell line, which precisely recapitulates this synergistic mechanism and provides an efficient assay platform for activity screening and mechanism-of-action validation of PD-1/IL-2–based therapeutics, thereby accelerating drug development.
