| Catalog Number | KC-1507 |
|---|---|
| Cell Line Name | Jurkat-NFAT- Luc2-CD16a-V158 Cell Line |
| Clone Number | 3# |
| Host Cell Line | Jurkat-NFAT-Luc2 cell line |
| Description | Jurkat cell line stable expressing exogenous luciferase gene under the control of NFAT responsive element and human FCGR3A amino acid bearing F158V mutation |
| Quantity | Two vials of frozen cells (≥2-106/vial) |
| Stability | Stable in culture over a minimum of 10 passages |
| Application | Drug screening and biological assays |
| Freezing Medium | 70% RPMI1640+20% FBS+10% DMSO |
| Propagation Medium | RPMI1640+10%FBS+300µg/mL Hygromycin B+0.75µg/mL Puromycin |
| Selection Marker | Puromycin, HygromycinB |
| Morphology | Lymphoblast |
| Subculture | Split saturated culture 1:4-1:6 every 2-3 days; seed out at about 1-3 × 105 cells/mL |
| Incubation | 37 °C with 5% CO2 |
| Storage | Liquid nitrogen immediately upon receiving |
| Doubling Time | Approximately 26 hours |
| Mycoplasma Status | Negative |
| In Vivo Validation | NA |
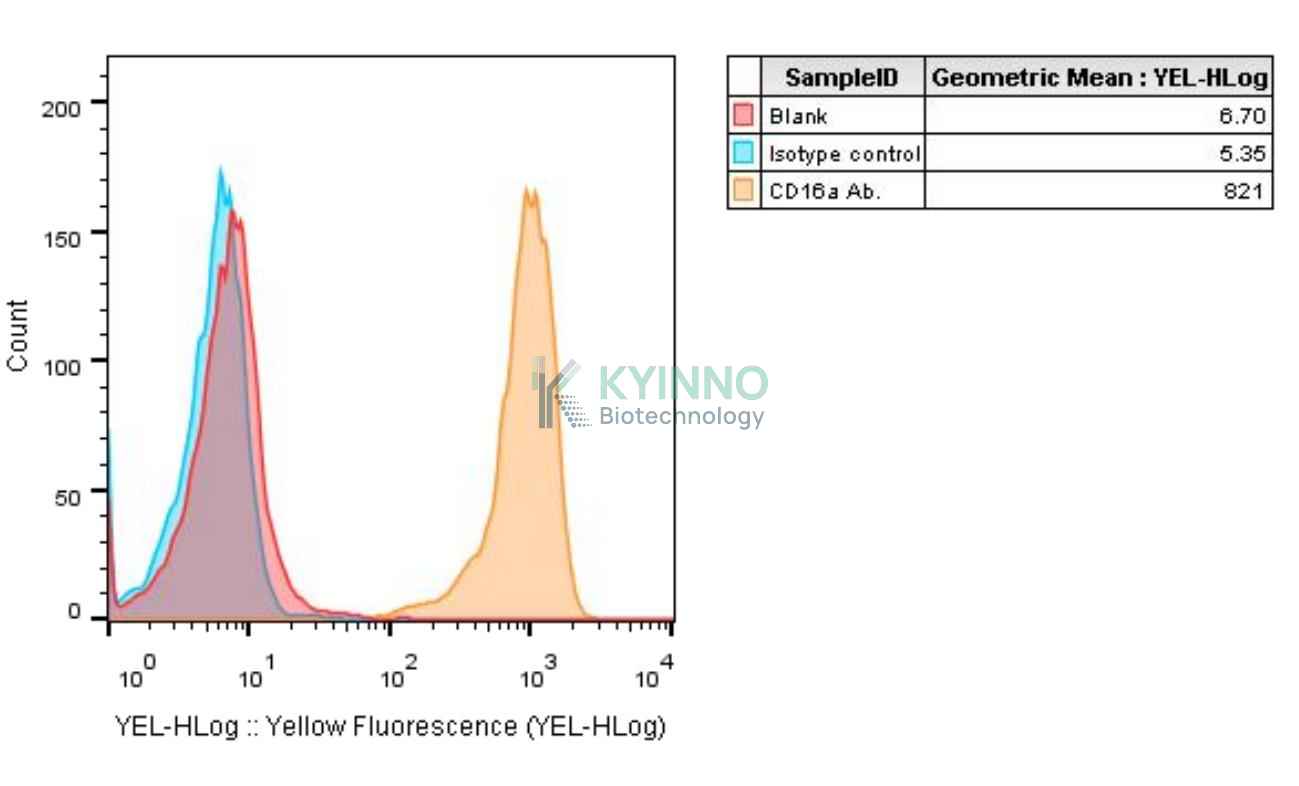
Figure 1: Characterization of CD16a-V158 overexpression in Jurkat NFAT-Luc2-CD16a-V158 reporter cell line using FACS.
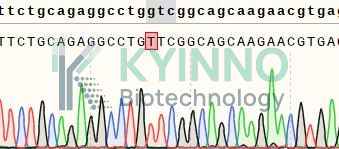
Figure 2: Characterization of CD16a-V158 overexpression in Jurkat NFAT-Luc2-CD16a-V158 reporter cell line using RT-PCR sequencing.